Here is our story
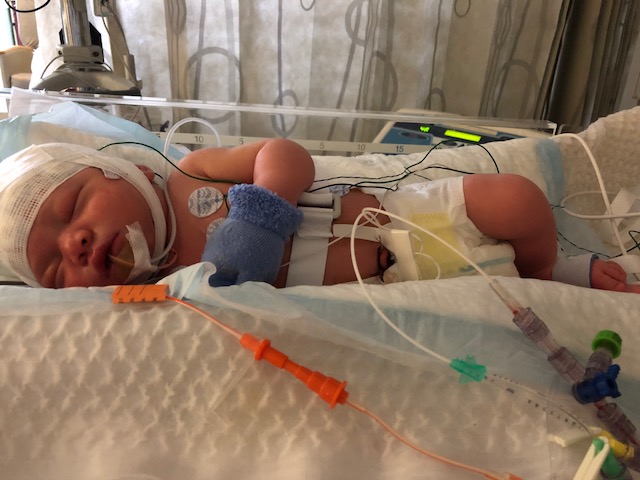
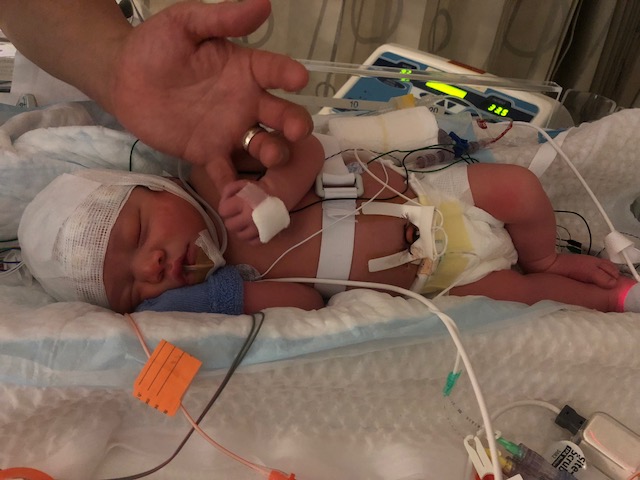
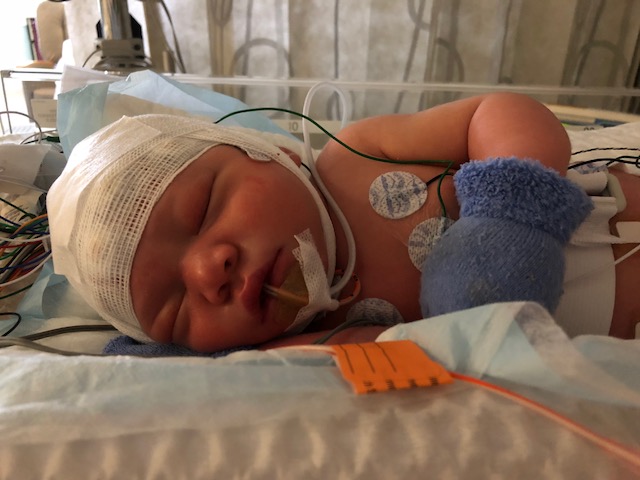
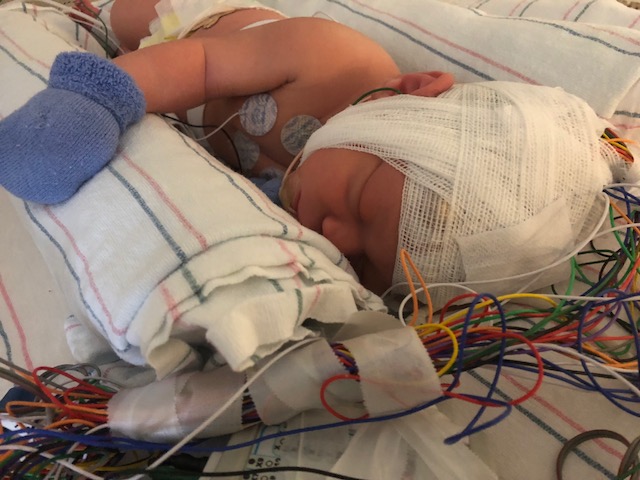
Hi there, some of you may know us and others may not. Thank you in advance for reading our story. My name is Patricia and my husband Ken and I just had a beautiful baby boy named “Julien” on July 14th 2018. When I was pregnant, many people told me that “it takes a village” to raise a child. I didn’t fully understand what that meant until now. We are reaching out to see if our “village” and “neighboring towns” would be willing to help us with the cost of a life-changing treatment for our baby Julien.
Here is our story:
On Friday July 13th, I went about my day as usual. I went to work, went out for lunch, etc. I noticed that I didn’t feel the baby move so I called my husband to let him know. I remember friends telling me that the baby doesn’t move as much at the very end of a pregnancy (especially if he is on the larger side like Julien was) so I assumed it was ok. I went home from work and drank a green tea latte which wasn’t something I normally drank while I was pregnant but I knew that the caffeine would make the baby move a little. Nothing. Ken got home from work and I told him that I was concerned so we called my midwife. She told me to come to Northside Hospital (which is where I was going to deliver anyway) and that she was on call and would see me there. When we arrived, they hooked me up to an IV and a heartrate and fetal movement monitor. His heartrate was very strong, however the fetal movement line was very flat. They put me on a sugar water IV to see if it would increase fetal movement and it didn’t. The decision was made to slowly induce me so that we could get the baby out without much stress. My midwife and nurse told my husband to go back home to pack his hospital bag (when we left the house we only grabbed my bag) but not to rush because we wouldn’t be having the baby for at least 12-16 hours